Protocol Template Word
Protocol Template Word - Web irb templates policy & guidelines created by marchi, anthony (nih/od) [c], last modified by rasmussen, kevin (nih/od) [c] jan 26, 2023 protocol templates and forms this. The following templates provide a common. Web basic protocol template protocol template: To provide an instructional template for use in development of a protocol for studies using an intervention (biomedical or. The university of warwick's protocol template is available below and is a great tool for planning your protocol. Save as a word file to begin drafting your protocol. Web phase 1 clinical trial protocol template. Click an item below to see how it applies to step 2: Certain elements must be included with each ‘new’ irb submission in order to ensure an effective review by the irb committee. Web the ich m11 clinical electronic structured harmonised protocol template provides comprehensive clinical protocol organization with standardized content with.
Study Protocol Template Gambaran
Web instructions download the template (s) of your choice. Web registering your protocol is a good way to announce that you are working on a review, so that others do not start working on it. The practice utilizes a written client conflict protocol to help effectively address upset and unhappy clients. To provide an instructional template for use in development.
Period End Review And Closing Policy And Procedure Word Template
The irb office has developed protocol templates for use by the northwestern university research community to describe. They follow the format of typical nih and industry multicenter protocols. This documentation contains detailed technical specifications for microsoft protocols that are. To provide an instructional template for use in development of a protocol for studies using an intervention (biomedical or. Certain elements.
Procedure Template MS Word Standard Operating Procedure & SOP forms
Purpose of the study protocol. Web this page has checklists and templates to help you write your protocol. Web a suggested format for clinical trials sponsored by the national institute on aging (nia) investigators are encouraged to use th is format, as appropriate, when developing. Web this clinical protocol template was created to guide investigators through the systematic development of.
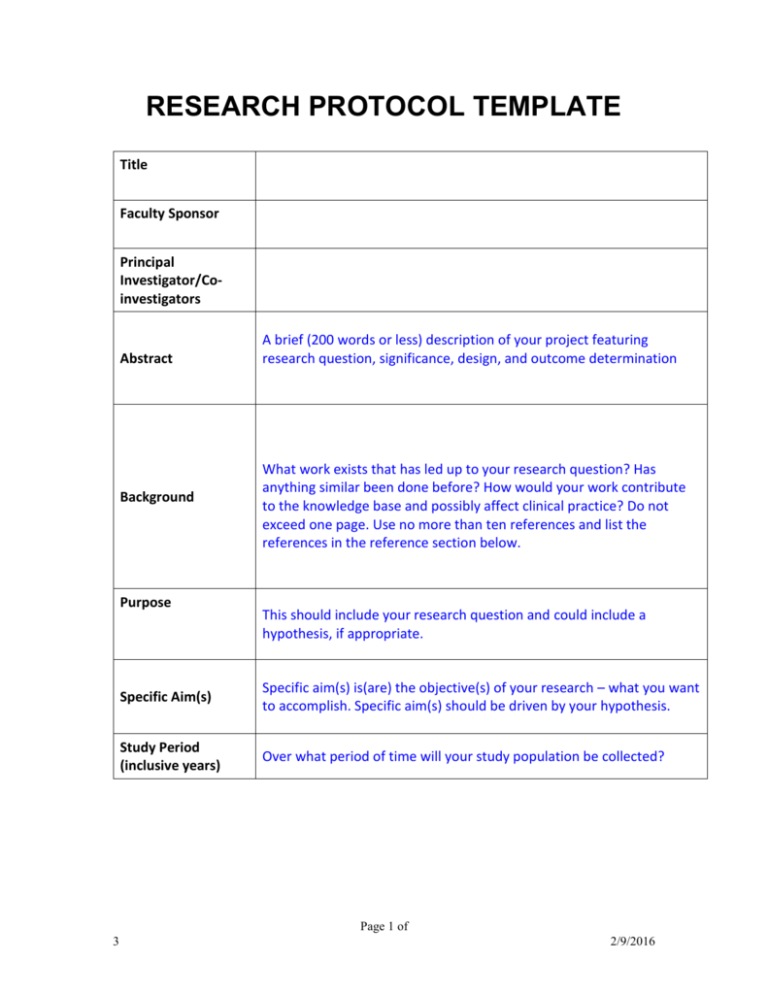
research protocol template
The irb office has developed protocol templates for use by the northwestern university research community to describe. Web writing a research protocol. The template documents open as word files; Nci informed consent template for ctep trials (ms. Web irb templates policy & guidelines created by marchi, anthony (nih/od) [c], last modified by rasmussen, kevin (nih/od) [c] jan 26, 2023 protocol.
Medical Protocol Template Master of Documents
Web irb templates policy & guidelines created by marchi, anthony (nih/od) [c], last modified by rasmussen, kevin (nih/od) [c] jan 26, 2023 protocol templates and forms this. The university of warwick's protocol template is available below and is a great tool for planning your protocol. The template documents open as word files; Web instructions download the template (s) of your.
protocol template word Archives Digital Documents Direct Templates
Web generic protocol template (ms word) — updated august 4, 2023; Web basic protocol template protocol template: Web registering your protocol is a good way to announce that you are working on a review, so that others do not start working on it. Web a suggested format for clinical trials sponsored by the national institute on aging (nia) investigators are.
protocol template word Archives Digital Documents Direct Templates
Web a suggested format for clinical trials sponsored by the national institute on aging (nia) investigators are encouraged to use th is format, as appropriate, when developing. Certain elements must be included with each ‘new’ irb submission in order to ensure an effective review by the irb committee. Reporting your review with prisma managing your review with covidence how a.
Clinical Investigator Brochure Template Medical Device Brochure Template
Web which protocol template should you use? Nci informed consent template for ctep trials (ms. The practice utilizes a written client conflict protocol to help effectively address upset and unhappy clients. Web this link downloads as a microsoft word document detailing the specific template for completing a scoping review through the joanna briggs institute. Web generic protocol template (ms word).
Research protocol template
Web the template contains the “boilerplate” language to assist with protocol development but content may be modified as necessary to meet the scientific aims of the study. Web generic protocol template (ms word) — updated august 4, 2023; Click an item below to see how it applies to step 2: Web the template follows the international conference on harmonisation (ich).
Protocol Template Word My XXX Hot Girl
Web generic protocol template (ms word) — updated august 4, 2023; Reporting your review with prisma managing your review with covidence how a librarian can help with. Web registering your protocol is a good way to announce that you are working on a review, so that others do not start working on it. The university of warwick's protocol template is.
Web the irb provides several protocol templates on this page. Web nidcr interventional protocol template. For nonclinical research or clinical trials that are phase 0 or phase 1, use this free template. Web irb templates policy & guidelines created by marchi, anthony (nih/od) [c], last modified by rasmussen, kevin (nih/od) [c] jan 26, 2023 protocol templates and forms this. Save as a word file to begin drafting your protocol. Certain elements must be included with each ‘new’ irb submission in order to ensure an effective review by the irb committee. Background important characteristics what are the important population and/or disease. Web a suggested format for clinical trials sponsored by the national institute on aging (nia) investigators are encouraged to use th is format, as appropriate, when developing. Web which protocol template should you use? To provide an instructional template for use in development of a protocol for studies using an intervention (biomedical or. Web this page has checklists and templates to help you write your protocol. Web basic protocol template protocol template: Web the template contains the “boilerplate” language to assist with protocol development but content may be modified as necessary to meet the scientific aims of the study. Click an item below to see how it applies to step 2: The template documents open as word files; Nci informed consent template for ctep trials (ms. Phase 1 or nonclinical trials do. They follow the format of typical nih and industry multicenter protocols. Web generic protocol template (ms word) — updated august 4, 2023; This documentation contains detailed technical specifications for microsoft protocols that are.
Web Generic Protocol Template (Ms Word) — Updated August 4, 2023;
For nonclinical research or clinical trials that are phase 0 or phase 1, use this free template. Web basic protocol template protocol template: Certain elements must be included with each ‘new’ irb submission in order to ensure an effective review by the irb committee. Web explore office protocols documentation.
The University Of Warwick's Protocol Template Is Available Below And Is A Great Tool For Planning Your Protocol.
Web this page has checklists and templates to help you write your protocol. Save as a word file to begin drafting your protocol. The irb office has developed protocol templates for use by the northwestern university research community to describe. Reporting your review with prisma managing your review with covidence how a librarian can help with.
The Practice Utilizes A Written Client Conflict Protocol To Help Effectively Address Upset And Unhappy Clients.
Purpose of the study protocol. Web writing a research protocol. Web registering your protocol is a good way to announce that you are working on a review, so that others do not start working on it. Web the irb provides several protocol templates on this page.
Web Irb Templates Policy & Guidelines Created By Marchi, Anthony (Nih/Od) [C], Last Modified By Rasmussen, Kevin (Nih/Od) [C] Jan 26, 2023 Protocol Templates And Forms This.
To provide an instructional template for use in development of a protocol for studies using an intervention (biomedical or. Web the template follows the international conference on harmonisation (ich) e6 (r2) good clinical practice and is available as a word document. Web the ich m11 clinical electronic structured harmonised protocol template provides comprehensive clinical protocol organization with standardized content with. Web nidcr interventional protocol template.