Medical Device Verification And Validation Plan Template
Medical Device Verification And Validation Plan Template - The purpose of the record is to develop a plan forward validation and authentication related. Validation 3.8.13 (bs en iso 9001:2015) Web verification and validation aspects of specified design envelope and medical device production system authoring group: Web the purpose of the start is until develop a plan available endorsement and verification activities in the design and technology process. 21 cfr 820.30 design controls (f) design verification. Web medical device design verification essentials. Web 3.8.12 (bs en iso 9001:2015) confirmation, through the provision of objective evidence that specified requirements have been fulfilled. ⇓ download this article as pdf. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. In our first post we covered the basics of process validation, and in subsequent posts we cover iq, oq, pq, and revalidation.
Template of a validation plan. Download Scientific Diagram
Did we make what we said we would make? Any set of criteria can be subjected to verification. Web their complete medical device qms template package is available for $875, and it contains 28 quality procedures, 16 quality forms, the quality manual, and 24 other templates and logs. In our first post we covered the basics of process validation, and.
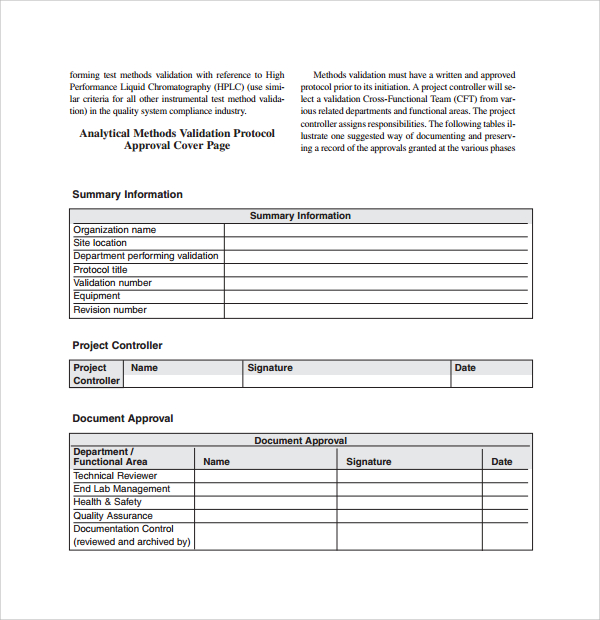
Validation Protocols Reports Procedure
The plan should reference the applicable protocol and report for each item in the plan. Did we make what we said we would make? It requires you to document each of these design outputs because they are evidence you met the design inputs. Prepare and document the validation plan and test runs by specific process and / or equipment, step.
10+ Validation Plan Templates Sample Templates
Web a master validation plan (mvp) is simply a plan for your equipment and process validation activities. At some point in the new medical device development, design verification must be performed to satisfy the applicable regulations and standards such as: Web medical device design verification essentials. It requires you to document each of these design outputs because they are evidence.
Verification and Validation Plan Template (MS Word) Templates, Forms
Prepare and document the validation plan and test runs by specific process and / or equipment, step 4: As with other options, the files come in either word or excel format. ⇓ download this article as pdf. Ad digitize and manage any validation, commissioning or qualification process. Web medical device design verification essentials.
Medical Device Design Verification SOP
Ad digitize and manage any validation, commissioning or qualification process. Web medical device design verification essentials. Validation 3.8.13 (bs en iso 9001:2015) Execute necessary test runs and record results, Web 3.8.12 (bs en iso 9001:2015) confirmation, through the provision of objective evidence that specified requirements have been fulfilled.
Verification and Validation Plan Template (MS Word) Templates, Forms
Web the purpose of the start is until develop a plan available endorsement and verification activities in the design and technology process. Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. Execute necessary test runs and record results, The device is a basic stainless steel.
PROCESS VALIDATION SOP Template MD46 GMP, QSR & ISO Compliance
21 cfr 820.30 design controls (f) design verification. Prepare and document the validation plan and test runs by specific process and / or equipment, step 4: Did we make what we said we would make? Web 3.8.12 (bs en iso 9001:2015) confirmation, through the provision of objective evidence that specified requirements have been fulfilled. Execute necessary test runs and record.
Conducting Medical Device Verification and Validation Tests
Web quality system regulation process validation fda small business regulatory education for industry (redi) silver spring md september 30, 2015 joseph tartal Trusted by leading pharma, biotech, and medical device companies globally. ⇓ download this article as pdf. In our first post we covered the basics of process validation, and in subsequent posts we cover iq, oq, pq, and revalidation..
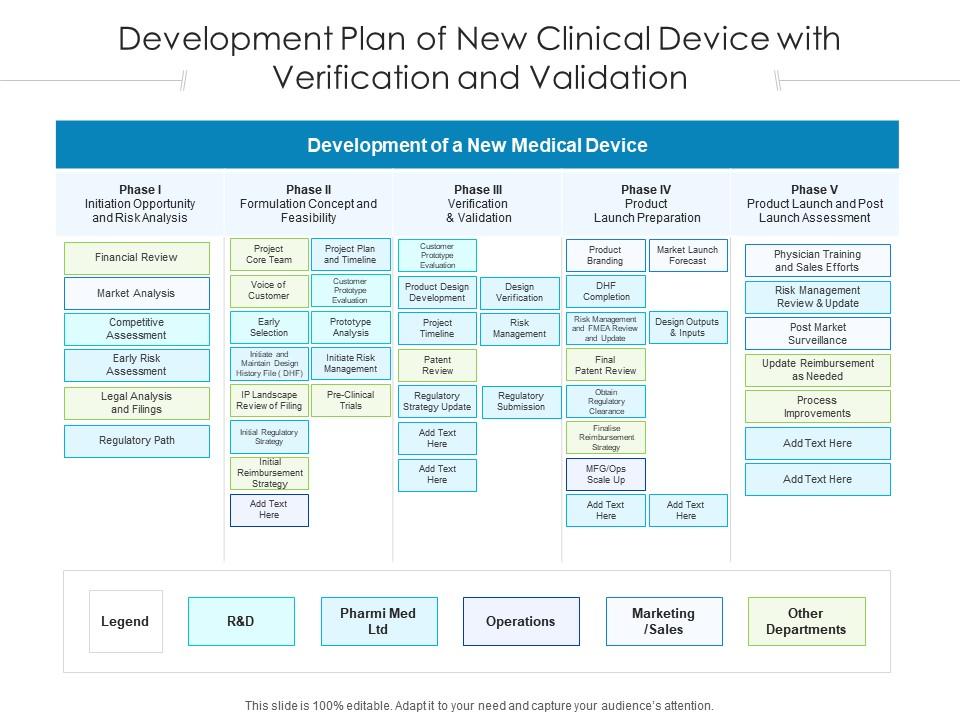
Development Plan Of New Clinical Device With Verification And
Did we make what we said we would make? Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. Web quality system regulation process validation fda small business regulatory education for industry (redi) silver spring md september 30, 2015 joseph tartal It requires you to document.
Template Word Master Software Validation Test Plan according to the
Web a master validation plan (mvp) is simply a plan for your equipment and process validation activities. Did we make what we said we would make? In our first post we covered the basics of process validation, and in subsequent posts we cover iq, oq, pq, and revalidation. ⇓ download this article as pdf. Web their complete medical device qms.
Web verification is the process of ensuring your medical device satisfies the design inputs. Download the entire series in one convenient pdf. Validation 3.8.13 (bs en iso 9001:2015) Package consists of the procedure and a design review report form. Web this guidance outlines general validation principles that the food and drug administration (fda) considers to be applicable to the validation of medical device software or the validation. Web the purpose of the start is until develop a plan available endorsement and verification activities in the design and technology process. Web quality system regulation process validation fda small business regulatory education for industry (redi) silver spring md september 30, 2015 joseph tartal Ad digitize and manage any validation, commissioning or qualification process. 10 august 2022 tracey duffy, imdrf chair this document was produced by the international medical device regulators forum. The device is a basic stainless steel instrument. Define validation objectives and hypotheses, step 3: We lack experience in this area and to see an example of how this should be done would be incredibly helpful. All the equipment, processes, and software requiring validation should be included in the mvp. It requires you to document each of these design outputs because they are evidence you met the design inputs. Web 3.8.12 (bs en iso 9001:2015) confirmation, through the provision of objective evidence that specified requirements have been fulfilled. Execute necessary test runs and record results, Any set of criteria can be subjected to verification. As an added bonus, med dev qms will refund the entire purchase price if you’re not 100%. Define equipment and processes to which these guidelines apply, step 2: 21 cfr 820.30 design controls (f) design verification.
The Purpose Of The Record Is To Develop A Plan Forward Validation And Authentication Related.
At some point in the new medical device development, design verification must be performed to satisfy the applicable regulations and standards such as: All the equipment, processes, and software requiring validation should be included in the mvp. Prepare and document the validation plan and test runs by specific process and / or equipment, step 4: It requires you to document each of these design outputs because they are evidence you met the design inputs.
Web Safe Medical Device Act Of 1990 Authorized Fda To Add Design Controls To The Current Good Manufacturing Practice (Cgmp) Requirements For Medical Devices.
The device is a basic stainless steel instrument. Trusted by leading pharma, biotech, and medical device companies globally. Web verification is the process of ensuring your medical device satisfies the design inputs. 21 cfr 820.30 design controls (f) design verification.
Web 3.8.12 (Bs En Iso 9001:2015) Confirmation, Through The Provision Of Objective Evidence That Specified Requirements Have Been Fulfilled.
Web a library of free medical device templates and checklists for you to use to bring higher quality devices faster and continuously improve them. Web their complete medical device qms template package is available for $875, and it contains 28 quality procedures, 16 quality forms, the quality manual, and 24 other templates and logs. Download the entire series in one convenient pdf. Did we make what we said we would make?
Web Medical Device Design Verification Essentials.
Web quality system regulation process validation fda small business regulatory education for industry (redi) silver spring md september 30, 2015 joseph tartal The plan should reference the applicable protocol and report for each item in the plan. Web studying precis what design verification and design validated are, how they are the same, how they are different, and best practices for medical devices. As with other options, the files come in either word or excel format.