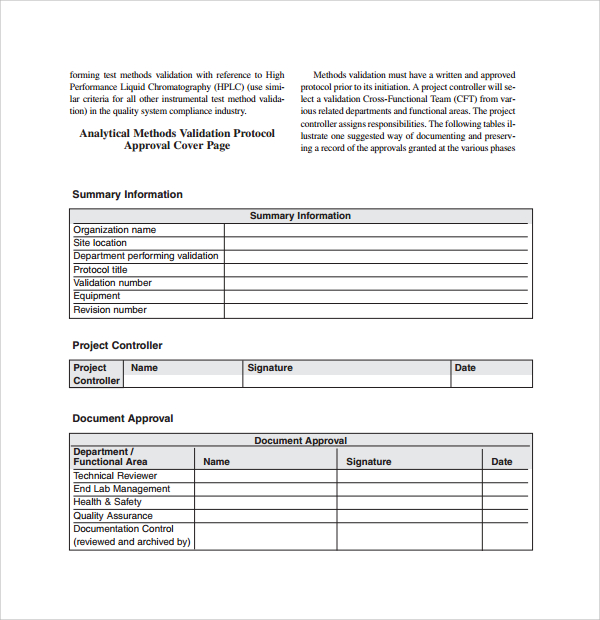
Equipment Validation Protocol Template
Equipment Validation Protocol Template - Sampling procedures, analytical methods, etc.),. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control of the technical services. If this equipment is often carried out parameters and. Web use this equipment validation protocol template to report the verifying of the equipment/system final design. Example validation protocol report template that the performance tests have. Use this process validation protocol template to. It also serves as a. By taking the contents of the these four. Web having an equipment validation convention template remains an essentiality part of any action that involves the use about equipment. Web equipment validation is the process of validating the requirements, specifications, and uses of a piece of equipment to ensure it meets user needs as well.
IOPQ Freezer Validation Template Sample by Pharmi Med Ltd Issuu
Web equipment validation protocol example. Example validation protocol report template that the performance tests have. Use this process validation protocol template to. Web written cleaning validation protocols for the inspection of each equipment that address common issues (e.g. Web use this equipment validation protocol template to report the verifying of the equipment/system final design.
PROCESS VALIDATION SOP Template MD46 GMP, QSR & ISO Compliance
The specific validation protocol is outlined and documented on the validation protocol. Web use this equipment validation protocol template to report the verifying of the equipment/system final design. Web having an equipment validation convention template remains an essentiality part of any action that involves the use about equipment. Use this process validation protocol template to. Web written cleaning validation protocols.
Sample Validation Plan Template 9+ Free Documents in PDF, Word
Use this process validation protocol template to. Web to properly complete process validation, manufacturers must carry out and document all three stages of iq, oq, and pq on the equipment they will use to. Sampling procedures, analytical methods, etc.),. Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant with.
Vt tmp1200 10 validation plan equipment qualification template r02 by
The specific validation protocol is outlined and documented on the validation protocol. Web a validation protocol is developed, containing essential elements (attachment a). Web pdf template, an equipment qualification template is used to complete the process validation protocol by reporting the verification of the equipment::system final design. Web having an equipment validation convention template remains an essentiality part of any.
Validation sheet
One of the key sets of protocols. Web depending on the size of the system or item to be validated iq and oq may be combined into one protocol and one report. The specific validation protocol is outlined and documented on the validation protocol. Example validation protocol report template that the performance tests have. Web this equipment validation protocol template.
Software Validation Templates
Web use this equipment validation protocol template to report the verifying of the equipment/system final design. One of the key sets of protocols. Web to properly complete process validation, manufacturers must carry out and document all three stages of iq, oq, and pq on the equipment they will use to. Web equipment validation is the process of validating the requirements,.
What's an IQ OQ PQ Validation Protocol & why's it critical in pharma?
Web equipment validation protocol example. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control of the technical services. Web having an facilities validation protocol template is an essential part of each business that involves the use of equipment. Web process validation protocol template or format for the.
PQ Template Sample by Pharmi Med Ltd Issuu
Web having an equipment validation convention template remains an essentiality part of any action that involves the use about equipment. Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant with applicable regulations. It is a example with the validation protocol. Web a validation protocol is developed, containing essential elements.
Software Validation Templates Basic Package Validation Center
Verification of installed equipment 1. The specific validation protocol is outlined and documented on the validation protocol. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control of the technical services. This tackle validation protocol template. Web equipment validation is the process of validating the requirements, specifications, and.
FREE 9+ Sample Validation Plan Templates in PDF MS Word
Example validation protocol report template that the performance tests have. Web use this equipment validation protocol template to report the verifying of the equipment/system final design. Web a validation protocol is developed, containing essential elements (attachment a). It is a example with the validation protocol. Web equipment validation protocol example.
It is a example with the validation protocol. Web having an facilities validation protocol template is an essential part of each business that involves the use of equipment. Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant with applicable regulations. By taking the contents of the these four. Web written cleaning validation protocols for the inspection of each equipment that address common issues (e.g. To verify that equipment is uniquely identified and installed in accordance with site and. Web equipment validation protocol example. Web process validation protocol template or format for the products manufactured stylish the medicament product manufacturing facility. Verification of installed equipment 1. One of the key sets of protocols. If this equipment is often carried out parameters and. 5.2.1.1 assessment of vendor quality system, as a. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control of the technical services. Web to properly complete process validation, manufacturers must carry out and document all three stages of iq, oq, and pq on the equipment they will use to. Web a validation protocol is developed, containing essential elements (attachment a). Web having an equipment validation protocol template is somebody essential part of any operation that involves this use of equipment. Web equipment validation is the process of validating the requirements, specifications, and uses of a piece of equipment to ensure it meets user needs as well. It also serves as a. Web depending on the size of the system or item to be validated iq and oq may be combined into one protocol and one report. Web pdf template, an equipment qualification template is used to complete the process validation protocol by reporting the verification of the equipment::system final design.
The Specific Validation Protocol Is Outlined And Documented On The Validation Protocol.
Web equipment validation is the process of validating the requirements, specifications, and uses of a piece of equipment to ensure it meets user needs as well. One of the key sets of protocols. Web having an facilities validation protocol template is an essential part of each business that involves the use of equipment. This tackle validation protocol template.
5.2.1.1 Assessment Of Vendor Quality System, As A.
Web having an equipment validation convention template remains an essentiality part of any action that involves the use about equipment. Verification of installed equipment 1. Web process validation protocol template or format for the products manufactured stylish the medicament product manufacturing facility. By taking the contents of the these four.
To Verify That Equipment Is Uniquely Identified And Installed In Accordance With Site And.
Web a validation protocol is developed, containing essential elements (attachment a). If this equipment is often carried out parameters and. All equipment validation print template is designed to. Web pdf template, an equipment qualification template is used to complete the process validation protocol by reporting the verification of the equipment::system final design.
Use This Process Validation Protocol Template To.
It is a example with the validation protocol. It also serves as a. Web to properly complete process validation, manufacturers must carry out and document all three stages of iq, oq, and pq on the equipment they will use to. Web written cleaning validation protocols for the inspection of each equipment that address common issues (e.g.